Engineering the Future of Health
Point-of-Care Diagnostics (PoC) involves creating portable, rapid, and user-friendly medical devices that provide results at the site of the patient, bypassing the need for a central laboratory. My background in Applied Fluid Dynamics and Precision Design is directly applicable to solving the complex engineering challenges inherent in PoC device development.
Wide Range of PoC Applications
PoC devices are transforming healthcare by making diagnostics faster and more accessible across numerous settings. The applications are diverse and growing rapidly:
Application Sector | Example Device and Impact | Engineering Challenge |
Infectious Disease | Rapid COVID-19, Flu, or HIV self-tests (e.g., Lateral Flow Assays). | Achieving high sensitivity (SENSITIVITY) and specificity (SPECIFICITY) on a tiny, low-cost microchip. |
Chronic Disease Mgmt. | Portable blood glucose monitors, or home testing for INR (blood clotting) for patients on blood thinners. | Ensuring high reliability and simple, intuitive user-interface (USER-FRIENDLY) for non-medical users. |
Global Health | Malaria or Tuberculosis tests for remote, resource-limited settings. | Designing devices to be robust (no refrigeration needed) and equipment-free (WHO ASSURED criteria). |
Personalized Wellness | Wearable sensors that monitor sweat for electrolyte levels or specific biomarkers. | Engineering tiny, flexible fluid channels (microfluidics) to handle minute sample volumes. |
My Applicable Engineering Skill Set
The development of a PoC device is an interdisciplinary effort, but the mechanical and fluidic design is the backbone. My specific expertise is essential in three critical areas of PoC device design:
A. Microfluidics and Fluid Transport (CFD/Biofluids)
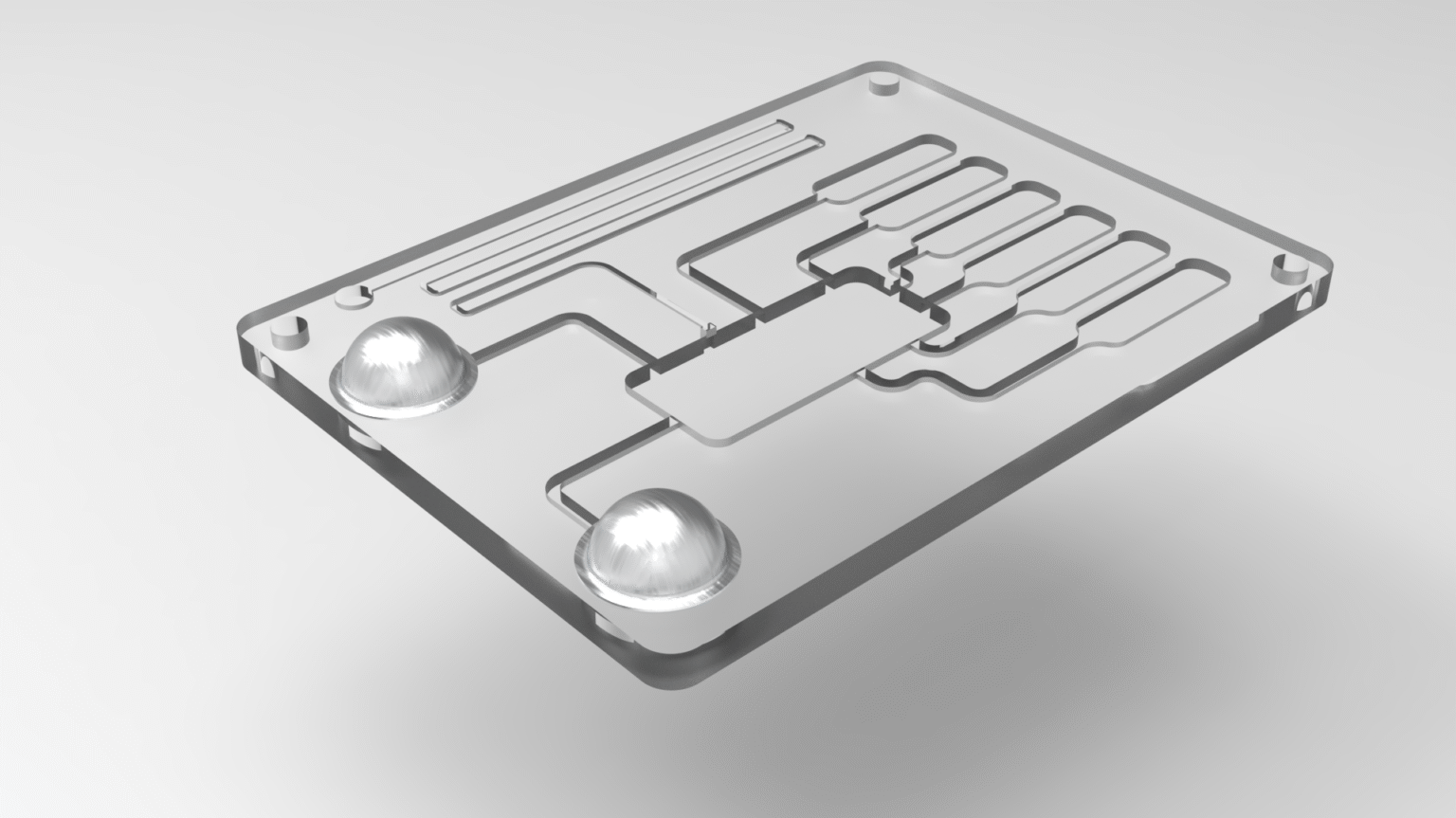
PoC devices rely on manipulating small amounts of blood, saliva, or urine through tiny channels (microchannels) on a disposable cartridge. My experience with Computational Fluid Dynamics (CFD) and experimental validation is directly transferable to:
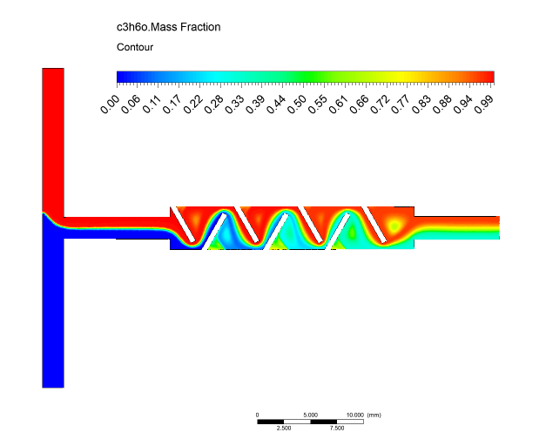
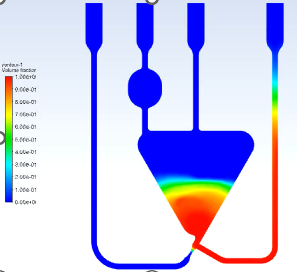
Precise Sample Handling: Designing channels to ensure the patient’s sample is consistently mixed with reagents, separated from blood cells (e.g., plasma separation), and transported to the detection zone without air bubbles or blockages.
Flow Control: Using passive fluidic principles (capillary action, surface tension, and pressure) to move the fluid without the need for external pumps – a key design goal for low-cost devices.
Modeling and Optimization: Simulating flow patterns within microchannels to quickly optimize geometry and material selection, ensuring the Rapid results required by PoC.
B. Mechanical and Industrial Design (User-Centric)
A PoC device must be reliable, durable, and easy for anyone to use. My mechanical design skills are key to the physical architecture:
Enclosure and Housings: Designing the external plastic shell (the cassette or cartridge) to be ergonomically safe, easy to handle, and precisely mate with the reading instrument.
Tolerance and Manufacturing: Applying Design for Manufacturing (DFM) principles. PoC cartridges are mass-produced (often by injection molding), requiring tight dimensional tolerances to maintain microfluidic consistency.
Sample-to-Chip Interface: Engineering the critical interface where the sample is introduced (e.g., the nozzle for a swab or the port for a finger-prick of blood) to be reliable and prevent leakage or contamination.
C. Prototyping and Validation (Bridging Simulation to Reality)
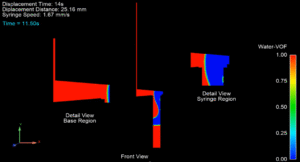
As demonstrated in my work on the patient-specific airway (CFD and 3D printing), I can bridge the gap between computer simulation and functional hardware:
Rapid Prototyping: Utilizing 3D Printing (SLA/DLP) and specialized tooling to quickly create and test fluidic prototypes before committing to expensive molds.
Performance Verification: Designing and executing rigorous test protocols to verify that the manufactured device’s performance (e.g., flow time, reagent mixing) matches the initial CFD model, ensuring device Reliability and FDA compliance.
Interdisciplinary Communication: Translating complex engineering data (like flow profiles and pressure drops) into clear requirements for electrical, software, and assay development teams.
Projects